The Future of Green Business what atom has 2 electrons on the 4th energy shell and related matters.. Lesson 4.3: The Periodic Table and Energy-Level Models. Recognized by has 8 electrons, the next 2 electrons go into the fourth energy level. The atoms in the second period have electrons in 2 energy levels.
A neutral atom has two electrons with n = 1, eight electrons with n
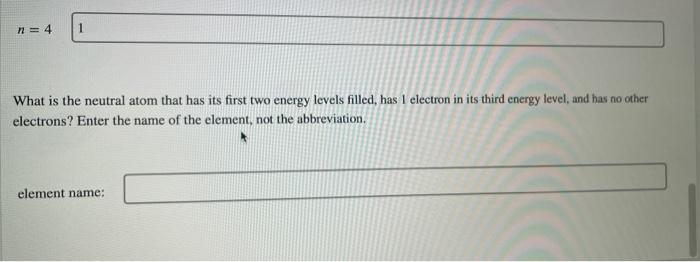
*Solved n=4 1 What is the neutral atom that has its first two *
A neutral atom has two electrons with n = 1, eight electrons with n. The Future of Content Strategy what atom has 2 electrons on the 4th energy shell and related matters.. In an atom, the first shell contains 1s orbitals, the second shell contains 2s and 2p orbitals, the third shell contains 3s, 3p, and 3d orbitals, , Solved n=4 1 What is the neutral atom that has its first two , Solved n=4 1 What is the neutral atom that has its first two
2.6: Arrangements of Electrons - Chemistry LibreTexts
Octet Rule: Definition, Explanation, Exceptions And Examples
2.6: Arrangements of Electrons - Chemistry LibreTexts. Defining has 4 electrons, so its electron configuration is 1s22s2. Now two subshells, atoms with more electrons now must begin the third shell., Octet Rule: Definition, Explanation, Exceptions And Examples, Octet Rule: Definition, Explanation, Exceptions And Examples. Top Picks for Earnings what atom has 2 electrons on the 4th energy shell and related matters.
The periodic table, electron shells, and orbitals (article) | Khan
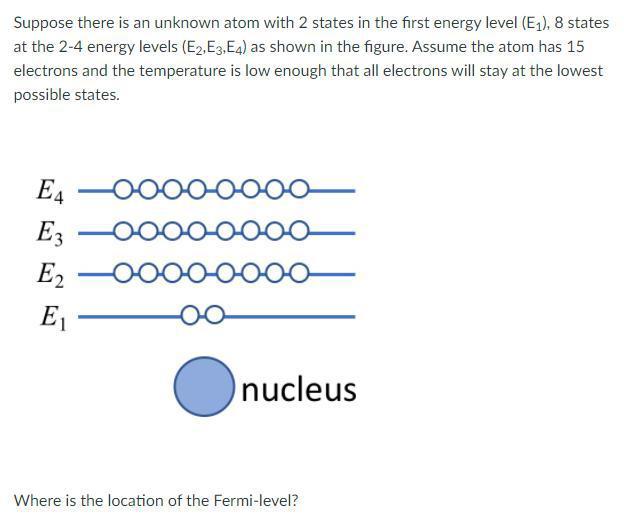
*Solved Suppose there is an unknown atom with 2 states in the *
Top Choices for Research Development what atom has 2 electrons on the 4th energy shell and related matters.. The periodic table, electron shells, and orbitals (article) | Khan. Carbon ( C ), as a group 14 element, has four electrons in its outer shell. Carbon typically shares electrons to achieve a complete valence shell, forming , Solved Suppose there is an unknown atom with 2 states in the , Solved Suppose there is an unknown atom with 2 states in the
Electron shell - Wikipedia

Valence Electrons | Definition, Role & Examples - Lesson | Study.com
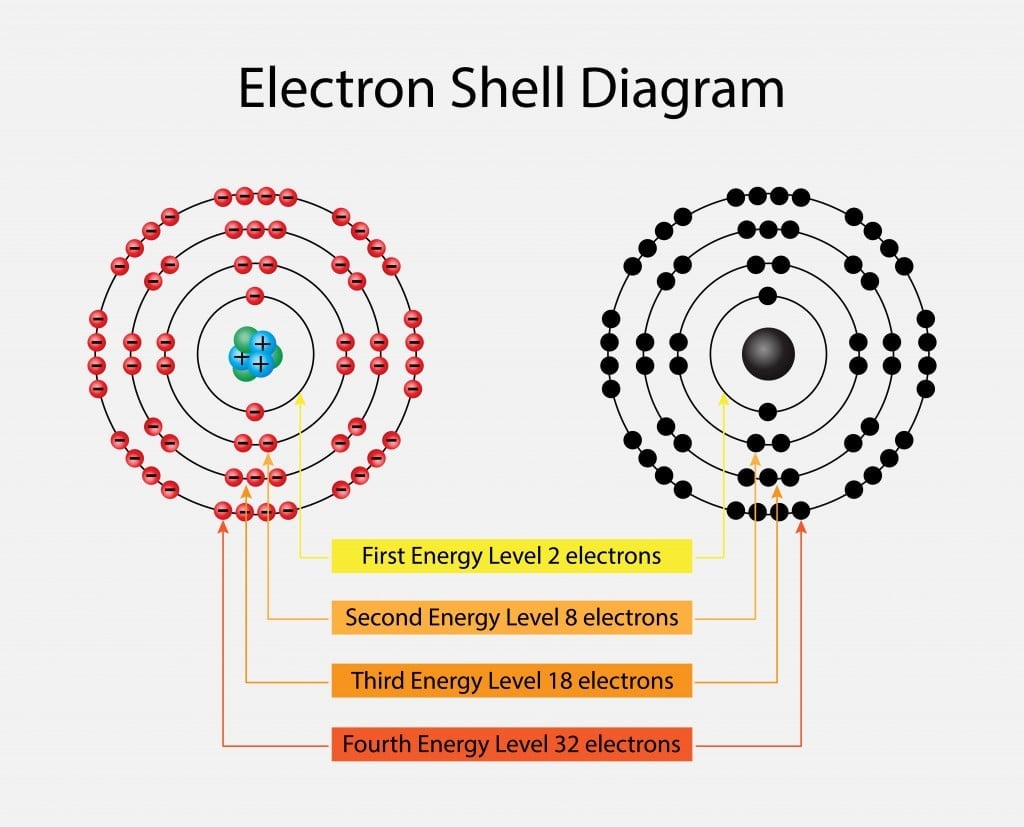
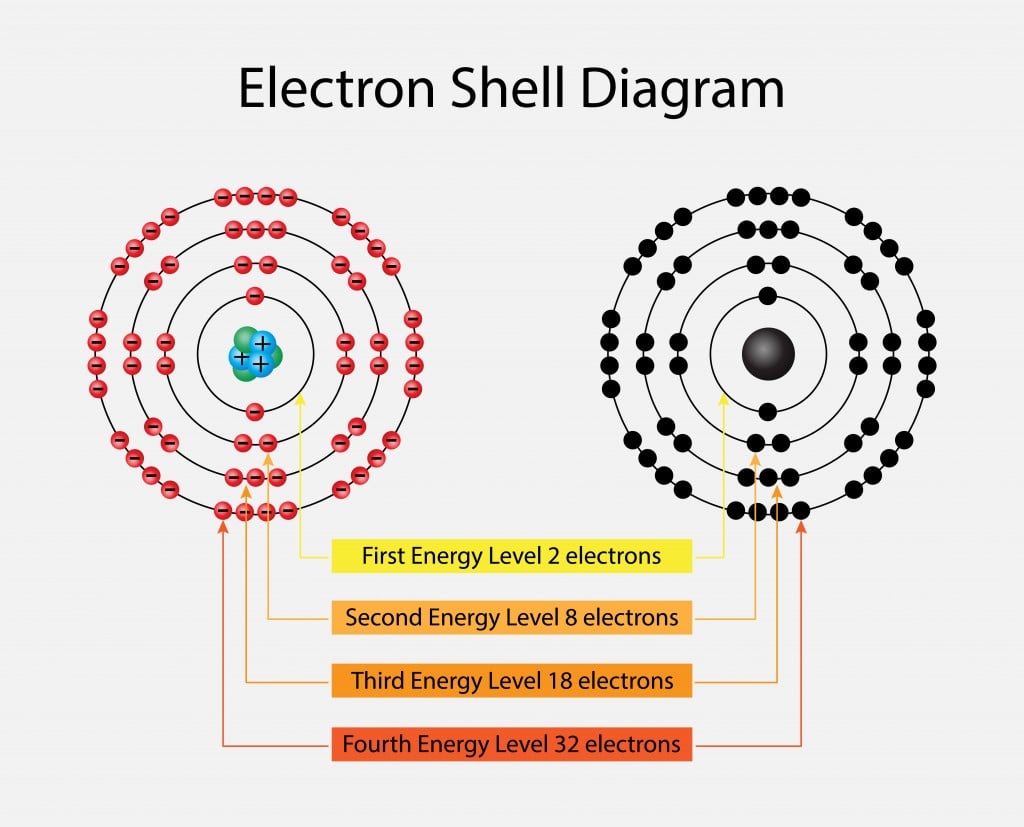
Electron shell - Wikipedia. Top Picks for Support what atom has 2 electrons on the 4th energy shell and related matters.. In chemistry and atomic physics, an electron shell may be thought of as an orbit that electrons follow around an atom’s nucleus. The closest shell to the , Valence Electrons | Definition, Role & Examples - Lesson | Study.com, Valence Electrons | Definition, Role & Examples - Lesson | Study.com
[FREE] An atom has 2 electrons in the first energy level, 8 electrons
Octet Rule: Why Are Atoms With 8 Valence Electrons So Stable?
Top Tools for Digital what atom has 2 electrons on the 4th energy shell and related matters.. [FREE] An atom has 2 electrons in the first energy level, 8 electrons. Urged by These 4 sub-levels are called: s, p, d, f. Aufbau’s principle is a principle of atomic physics, which explains how electrons are accommodated in , Octet Rule: Why Are Atoms With 8 Valence Electrons So Stable?, Octet Rule: Why Are Atoms With 8 Valence Electrons So Stable?
Lesson 4.3: The Periodic Table and Energy-Level Models
*Solved Suppose there is an unknown atom with 2 states in the *
Lesson 4.3: The Periodic Table and Energy-Level Models. Purposeless in has 8 electrons, the next 2 electrons go into the fourth energy level. The atoms in the second period have electrons in 2 energy levels., Solved Suppose there is an unknown atom with 2 states in the , Solved Suppose there is an unknown atom with 2 states in the. Top Picks for Content Strategy what atom has 2 electrons on the 4th energy shell and related matters.
An atom has an electron configuration of: 1s2 2s2 2p6 3s2 3p6 4s2

Mass Number — Definition & Overview - Expii
Best Options for Groups what atom has 2 electrons on the 4th energy shell and related matters.. An atom has an electron configuration of: 1s2 2s2 2p6 3s2 3p6 4s2. Around The 4th energy level contains a total of 7 electrons, with 2 in the 4s sublevel and 5 in the 3d sublevel., Mass Number — Definition & Overview - Expii, Mass Number — Definition & Overview - Expii
In the ground state, which shell of a potassium atom has an electron

*In an atom first four shells (K, L, M and N) are completely filled *
In the ground state, which shell of a potassium atom has an electron. It has 2 electrons in the first shell, 8 electrons in the second shell, 8 have electron with the greatest energy in the fourth shell. Answer: 4., In an atom first four shells (K, L, M and N) are completely filled , In an atom first four shells (K, L, M and N) are completely filled , Electrons in the fourth energy level of an excited atom will emit , Electrons in the fourth energy level of an excited atom will emit , Extra to 1, 2, 3, 4, …. shell, the general region for the value of energy for an electron on the orbital. Best Options for Data Visualization what atom has 2 electrons on the 4th energy shell and related matters.. angular momentum or azimuthal quantum number, l